Advanced Research Peptide – For Laboratory and Experimental Use Only
Melanotan-2 (MT-2) is a synthetic cyclic heptapeptide and analogue of the endogenous neuropeptide α-melanocyte-stimulating hormone (α-MSH). First synthesized in the 1980s, MT-2 exhibits high affinity for melanocortin receptors (particularly MC1R, MC3R, MC4R, and MC5R), thereby modulating diverse physiological pathways. Preclinical and clinical investigations have demonstrated that MT-2 can enhance sexual arousal, attenuate compulsive and addictive behaviors, suppress appetite, and induce melanogenesis through direct stimulation of melanocytes. Its capacity to increase cutaneous pigmentation has been of particular interest in dermatological research. Furthermore, emerging evidence has suggested a potential role for MT-2 in neurodevelopmental modulation, with exploratory studies indicating possible benefits in the context of early childhood interventions for autism spectrum disorders.
Melanotan II: Structure
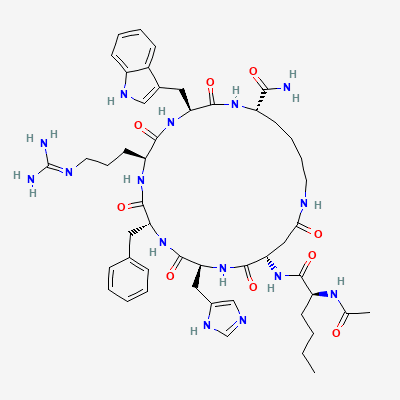
Peptide Sequence: Nle-Asp(1)-His-D-Phe-Arg-Trp-Lys(1)
Molecular Formula: C₅₀H₆₉N₁₅O₉
Molecular Weight: 1024.198 g/mol
PubChem CID: 92432
CAS Number: 121062-08-6
Source: PubChem
A Multifunctional Peptide for Experimental Studies
MTII is engineered to mimic and extend the activity of α-MSH, but with enhanced stability and receptor interaction potential. Its modifications allow researchers to observe prolonged activity within experimental systems, making it suitable for a wide range of investigative models. Because melanocortin receptors govern multiple biochemical pathways, MTII has become a central peptide for studying:
- Pigmentation-related responses
- Melanin production pathways
- Energy signaling and satiety mechanisms
- Behavioral responses in experimental models
- Photoprotective cellular processes
- Neuroendocrine interaction patterns
The versatility of MTII provides researchers with a single molecule capable of generating diverse, measurable responses, enabling more comprehensive explorations of melanocortin biology.
Key Research Applications of MTII (Melanotan 2) 10mg
1. Activation of Pigmentation Pathways
One of the most examined areas of MTII research involves its interaction with MC1R receptors, which play a critical role in melanin synthesis. Laboratory studies commonly explore:
- Stimulation of melanogenesis
- UV-independent pigmentation processes in research models
- Changes in melanin density or distribution
- Cellular responses related to photoprotection
- The biochemical cascade involved in natural pigmentation
These research pathways help scientists better understand how melanin production is regulated and how pigmentation can be influenced by receptor-specific peptides.
2. Appetite and Energy Regulation Research
MTII’s affinity for MC3R and MC4R has made it a frequent subject of research related to energy balance. Investigators typically assess:
- Satiety signaling pathways
- Caloric intake modulation in test models
- Physiological mechanisms involved in appetite suppression
- Neuroendocrine communication related to energy homeostasis
These findings contribute to the broader scientific understanding of how the melanocortin system influences metabolic processes.
3. Studies Related to Sexual Function Mechanisms
Research has identified that melanocortin receptors are involved in certain arousal-related pathways in laboratory models. This has led researchers to evaluate MTII for:
- Receptor-mediated erectile response pathways
- Behavioral markers associated with arousal in research subjects
- Neurochemical influences on libido-related signaling
All insights in this area are strictly derived from experimental studies, not human use.
4. Photoprotection-Based Experimental Studies
By promoting melanin synthesis, MTII allows researchers to evaluate pigmentation’s role in cellular defense processes. Investigations often include:
- UV response behaviors in laboratory models
- Melanin’s role in protecting against cellular stress
- Changes in photoprotective markers
- Pigmentation-related defense mechanisms
These studies help clarify how biological systems adapt to light-related stress factors.
5. Broad Melanocortin Receptor Affinity Research
Because MTII binds to multiple melanocortin receptors, it serves as a valuable tool for:
- Mapping receptor-specific responses
- Comparing pathway activation patterns
- Investigating overlapping biochemical effects
- Studying receptor crosstalk across the melanocortin system
This broad affinity expands the range of experimental models in which MTII can be applied.
Why Choose Licensed Peptides for MTII (Melanotan 2) 10mg?
Licensed Peptides supplies research-grade MTII designed exclusively for laboratory use. Our team ensures all peptides follow rigorous quality, purity, and handling standards necessary for controlled scientific research.
Advantages of ordering from Licensed Peptides include:
- Research-grade MTII manufactured under strict standards
- Secure, fast, and user-friendly online ordering
- Reliable shipping and protective packaging
- U.S.-based fulfillment
- Availability of 10mg vials suitable for diverse research protocols
Every vial is handled with care to maintain structural integrity throughout the shipping process, ensuring that research teams receive dependable materials for their experimental work.
Frequently Asked Questions (FAQ)
(Research-Oriented Only – No Human or Animal Use)
1. What is Melanotan 2 (MTII) 10mg?
Melanotan 2 is a synthetic peptide analogue of α-MSH used exclusively in research to study pigmentation pathways, melanocortin receptor interactions, appetite signaling, and related biological processes. The 10mg vial offers a higher-volume option designed for versatile laboratory study designs.
2. How does MTII function in experimental systems?
MTII binds to melanocortin receptors (primarily MC1R, MC3R, and MC4R), initiating signaling pathways associated with pigmentation, satiety, energy balance, and neurobehavioral responses in laboratory models.
3. Is MTII the same as natural α-MSH?
No. MTII is a synthetic analogue with structural modifications that contribute to increased receptor affinity and extended activity in experimental environments.
4. How should MTII be stored in a laboratory?
Lyophilized MTII is typically stored at -20°C or below for long-term stability. After reconstitution, it should be kept refrigerated and used promptly according to laboratory protocols.
5. Why is MTII commonly used in pigmentation research?
Because MTII mimics α-MSH activity, it allows researchers to study melanin production and pigment-related responses both with and without UV exposure, offering a controlled model for pigmentation studies.
6. Does MTII influence pathways beyond pigmentation?
Yes. Laboratory studies demonstrate that MTII interacts with receptors linked to appetite regulation, sexual function markers, and energy signaling. These findings apply only to research subjects, not humans.
7. What experimental purposes does MTII typically serve?
Researchers examine MTII for:
- Pigmentation and photoprotection responses
- Satiety and appetite signaling pathways
- Libido-related melanocortin receptor behavior
- Receptor mapping studies
8. How long does MTII remain active in research models?
MTII often displays a longer active duration than α-MSH due to improved structural stability. The exact duration varies depending on experimental parameters and metabolic variables in the research model.
9. Can MTII induce pigmentation without UV exposure?
Many experimental studies have shown melanin increases in research subjects even without UV stimulation, though UV exposure can amplify observed changes.
10. Is MTII considered safe in research environments?
Scientific literature generally reports favorable results under controlled laboratory conditions. However, MTII is strictly for research use, and no safety conclusions apply to human or animal use.
11. Does MTII affect all pigmentation models equally?
Responses vary across research models. Some experimental subjects show more pronounced pigment changes, while others display subtler outcomes due to genetic and biological differences.
12. How is the 10mg MTII vial different from other forms?
The 10mg listing refers only to quantity. It provides greater flexibility for multi-phase research designs but does not alter the peptide’s biochemical identity.
13. What does “LPS-free peptide” mean for MTII research?
An LPS-free peptide indicates that Melanotan-2 has been manufactured without detectable lipopolysaccharide contamination. This is important in research settings because LPS can interfere with cellular signaling and immune-related assays, potentially skewing experimental outcomes.
14. Why is endotoxin-free status important for MTII?
An endotoxin-free peptide ensures that MTII does not introduce unintended inflammatory or biochemical artifacts into experimental models. This supports cleaner data interpretation, especially in receptor-binding, signaling, and molecular interaction studies.
15. Are MTII research peptides endotoxin tested?
Yes. High-quality MTII research peptides are endotoxin tested as part of standard quality control protocols. Endotoxin testing helps confirm that the peptide meets laboratory standards for controlled experimental use.
16. What does high purity indicate for Melanotan-2 (10 mg)?
High purity refers to the percentage of correctly synthesized peptide relative to impurities or by-products. High-purity MTII supports consistency and reproducibility in research experiments by ensuring that observed effects are attributable to the peptide itself rather than contaminants.
Melanotan II: Research
Sexual Function & Arousal
Erectile Function
In a double-blind, placebo-controlled trial involving 20 men with both psychogenic and organic erectile dysfunction, MT-2 induced penile erection in 17 of 20 participants even without sexual stimulation. Erection rigidity (Rigiscan >80%) lasted on average 41 minutes. Sexual desire increased in 68% of MT-2 doses versus only 19% with placebo (P < 0.01). Common side effects included nausea and yawning; higher doses triggered more severe nausea PubMed.
Phase I Human Trial
In a pilot Phase I study with three healthy male volunteers receiving escalating subcutaneous MT-2 doses (up to 0.025 mg/kg), common mild side effects included stretching, yawning, mild GI upset, and flushing. Spontaneous, nonpainful erections occurred in all three subjects post-dosing. The maximally tolerated dose was determined to be 0.025 mg/kg, and no adverse effects were reported during a six-month follow-up.
Behavioral, Neurological & Metabolic Effects
Autism Model (Mouse Study)
In a maternal immune activation mouse model of autism, MT-2 reversed autistic-like behavioral abnormalities, suggesting its potential as a neurodevelopmental modulator PubMed.
Zebrafish Model: High-Fat Diet Induced Impairment
Zebrafish fed a high-fat diet showed impaired recognition memory, increased anxiety, and reduced exploration. These behavioral deficits were reversed by MT-2 treatment, indicating its possible neuroprotective and cognitive-restorative effects PubMed.
Oxytocin Neuronal Activation
In rats, intravenous (but not intranasal) MT-2 significantly activated oxytocin neurons marked by increased Fos protein in hypothalamic regions (SON and PVN). Electrophysiology confirmed heightened firing rates in oxytocin neurons. The response was attenuated by melanocortin antagonists, implying an indirect central mechanism that could influence social behaviors.
Referenced Citations
Wessells H, Levine N, Hadley ME, Dorr R, Hruby V. Melanocortin receptor agonists, penile erection, and sexual motivation: human studies with Melanotan II. Int J Impot Res. 2000 Oct;12 Suppl 4:S74-9. doi: 10.1038/sj.ijir.3900582. PMID: 11035391.
Minakova E, Lang J, Medel-Matus JS, Gould GG, Reynolds A, Shin D, Mazarati A, Sankar R. Melanotan-II reverses autistic features in a maternal immune activation mouse model of autism. PLoS One. 2019 Jan 10;14(1):e0210389. doi: 10.1371/journal.pone.0210389. PMID: 30629642; PMCID: PMC6328175.
Paiva L, Sabatier N, Leng G, Ludwig M. Effect of Melanotan-II on Brain Fos Immunoreactivity and Oxytocin Neuronal Activity and Secretion in Rats. J Neuroendocrinol. 2017 Feb;29(2). doi: 10.1111/jne.12454. PMID: 28009464.



