Overview
A Selective Growth Hormone – Releasing Peptide for Controlled Research Applications
Ipamorelin is a synthetic pentapeptide developed as a potent growth hormone (GH) secretagogue. It stimulates GH release both in vitro and in vivo with similar effectiveness to GHRP-6 but shows remarkable selectivity. Unlike other GH secretagogues such as GHRP-2 and GHRP-6, ipamorelin does not increase cortisol or ACTH levels, even at very high doses, and has no effect on other pituitary hormones like FSH, LH, prolactin, or TSH. This unique profile makes it the first GHRP-receptor agonist to display GH selectivity comparable to that of natural GHRH, highlighting its potential for future clinical development.
Ipamorelin : Structure
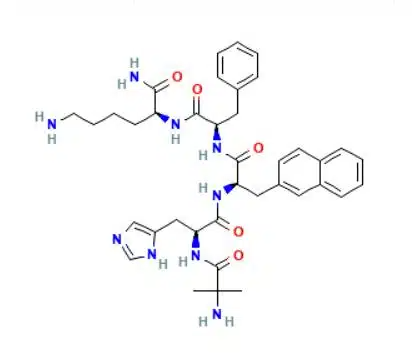
Peptide Sequence: Aib-His-D-2Nal-D-Phe-Lys
Molecular Formula: C₃₈H₄₉N₉O₅
Molecular W: 9831659
CAS Number: 170851-70-4
Source: PubChem
Mechanism of Action in Research Settings
Ipamorelin exerts its effects by binding to the GHS-R1a receptor, the same receptor activated by the endogenous peptide ghrelin. Upon receptor activation, a signaling cascade is initiated that prompts the pituitary gland to release growth hormone in short, discrete bursts. Importantly, research data suggests that Ipamorelin does not significantly amplify ghrelin-related appetite signaling, separating it from compounds such as GHRP-6 that strongly activate feeding-related pathways.
This receptor specificity enables researchers to isolate GH-centric responses in experimental models. Because growth hormone interacts with numerous downstream pathways, including IGF-1 expression, collagen synthesis signaling, lipid metabolism processes, and cellular turnover, Ipamorelin is frequently utilized to study how subtle modulation of GH pulses can influence broader biological systems without artificially elevating hormone levels beyond expected physiological ranges.
Why Ipamorelin Stands Out in Peptide Research
1. Highly Selective GH Release
Ipamorelin demonstrates a strong preference for growth hormone release without measurably increasing ACTH, cortisol, or prolactin in laboratory observations. This selectivity reduces hormonal cross-interference, allowing cleaner data interpretation in endocrine research.
2. Alignment With Circadian GH Rhythms
Growth hormone secretion is known to follow circadian patterns, with significant pulses occurring during rest phases. Ipamorelin’s pulsatile signaling profile makes it particularly useful for studies examining sleep-linked endocrine rhythms and GH-dependent repair cycles in controlled models.
3. Lipid Metabolism Research Applications
GH is closely associated with lipolysis pathways. Research involving Ipamorelin often explores how GH pulse enhancement may influence fat mobilization signaling, particularly during fasting or low-insulin states in experimental settings.
4. Connective Tissue and Structural Protein Signaling
Elevated GH and downstream IGF-1 signaling have been associated with collagen synthesis and connective tissue turnover. Ipamorelin is frequently referenced in studies examining tendon, ligament, and extracellular matrix remodeling pathways.
5. Cellular Aging and Regeneration Research
Because growth hormone plays a role in cell renewal and protein synthesis, Ipamorelin is commonly investigated in research related to aging mechanisms, oxidative stress modulation, and tissue regeneration signaling.
Documented Research Observations
Across laboratory and preclinical research contexts, Ipamorelin has been associated with:
- Increased endogenous GH pulse frequency and amplitude
- Minimal activation of stress-related hormonal pathways
- Downstream IGF-1 signaling modulation
- Altered lipid metabolism signaling markers
- Enhanced markers associated with cellular repair processes
It is important to note that these observations are derived strictly from controlled research environments and are used to understand biological mechanisms rather than to imply direct application.
Ipamorelin Research Profile Summary
- Selective growth hormone secretagogue activity
- Minimal influence on cortisol or prolactin pathways
- Supports investigation of endogenous hormone rhythm regulation
- Frequently studied in regeneration and recovery signaling models
- Useful for long-term, low-interference endocrine research
- Favorable stability profile for laboratory handling
Why Choose Focused Peptides for Ipamorelin Research Materials?
Focused Peptides is dedicated to supplying high-purity research peptides intended strictly for scientific and educational investigation. Based in the United States, our operations emphasize reliability, consistency, and quality assurance throughout every stage of fulfillment.
Each order is processed with secure packaging protocols designed to preserve compound integrity from dispatch through delivery. Our streamlined ordering system provides a smooth, encrypted checkout experience using major payment methods, ensuring efficient acquisition of research materials without unnecessary delays.
Focused Peptides does not market or sell products for human or animal use. All materials provided are labeled and intended exclusively for laboratory research, analytical testing, and educational purposes.
Frequently Asked Questions (FAQ)
What is Ipamorelin?
Ipamorelin is a synthetic growth hormone–releasing peptide used in laboratory studies to examine endogenous GH secretion mechanisms without significantly stimulating unrelated hormonal pathways.
How does Ipamorelin differ from earlier GHRPs in studies?
Research shows Ipamorelin exhibits greater receptor selectivity, resulting in GH release with minimal activation of cortisol, prolactin, or appetite-associated pathways.
Does Ipamorelin activate ghrelin pathways strongly?
While it binds to the ghrelin receptor, studies indicate that Ipamorelin does not strongly engage feeding-related signaling compared to other peptides in the same class.
Can Ipamorelin be combined with other research peptides?
Yes. In laboratory settings, Ipamorelin is often studied alongside GHRH analogs such as CJC-1295 (No DAC) or Sermorelin to evaluate synergistic GH pulse dynamics.
What biological systems are commonly studied using Ipamorelin?
Research commonly focuses on endocrine signaling, IGF-1 modulation, lipid metabolism pathways, connective tissue turnover, and cellular regeneration markers.
How quickly are changes observed in research models?
GH pulse modulation can be observed relatively quickly after exposure in experimental systems, while downstream signaling changes typically appear over longer research durations.
Is Ipamorelin linked to fat metabolism signaling in studies?
Yes. Growth hormone is known to influence lipid mobilization pathways, and Ipamorelin is frequently used to explore this relationship in controlled environments.
Does Ipamorelin influence sleep-related hormone cycles?
Research examining circadian GH secretion often includes Ipamorelin due to its alignment with naturally timed GH pulses observed during rest phases.
What side effects are noted in research observations?
Studies report minimal off-target hormonal activity. Observed effects are generally limited to transient GH-related signaling shifts rather than systemic disruptions.
Why is Ipamorelin considered “clean” in research literature?
Its reputation comes from its narrow receptor activity and minimal hormonal spillover, allowing investigators to study GH modulation without significant confounding endocrine variables.
How long are Ipamorelin research cycles typically conducted?
Research durations often range from 8–12 weeks, though longer-term protocols are documented depending on experimental design objectives.
Is Ipamorelin associated with fluid balance changes in studies?
Some GH-related signaling changes can influence water retention markers, though these effects are typically mild and stabilize over continued observation periods.
Why is Ipamorelin described as an LPS-free peptide in research settings?
An LPS-free peptide ensures the absence of lipopolysaccharide contaminants, which can activate inflammatory or immune signaling pathways and compromise experimental data. This is particularly important in endocrine and cellular signaling research where assay sensitivity is high.
What is the importance of using an endotoxin-free peptide for Ipamorelin studies?
An endotoxin-free peptide minimizes unintended biological interference, allowing researchers to attribute observed signaling changes specifically to GHS-R1a activation rather than contamination-induced artifacts.
How are research peptides endotoxin tested for Ipamorelin?
Research peptides endotoxin tested for Ipamorelin typically undergo standardized analytical assays (such as LAL testing) to confirm endotoxin levels fall within acceptable laboratory research thresholds.
Does high purity matter when working with Ipamorelin in laboratory research?
Yes. High-purity Ipamorelin supports experimental reproducibility by reducing batch variability and eliminating secondary peptide fragments that could otherwise influence receptor signaling outcomes.
Ipamorelin: Research
Ghrelin Receptor
The ghrelin receptor, a 7-transmembrane protein, is found at elevated levels in certain cancers and in heart failure, making it a valuable biomarker for imaging applications. Past PET tracers have mostly been limited to small-molecule quinalizonones and ghrelin-derived peptides. In this work, a new set of 4-fluorobenzoylated growth hormone secretagogues (GHSs) was designed and tested, spanning both peptide-based (GHRP-1, GHRP-2, GHRP-6) and peptidomimetic (G-7039, [1-Nal4]G-7039, ipamorelin) structures. The peptidomimetic analogue G-7039 proved to be the most promising scaffold for fluorine-18 incorporation. Its modified derivative, [1-Nal4,Lys5(4-FB)]G-7039, showed strong receptor affinity (IC50 = 69 nM) and potent in vitro activity (EC50 = 1.1 nM). Using N-succinimidyl-4-[18F]fluorobenzoate ([18F]SFB) for labeling, the radiotracer [1-Nal4,Lys5(4-[18F]-FB)]G-7039 was obtained with high yield (48%), excellent purity (≥99%), and good molar activity (>34 GBq/μmol). This radiolabeled compound represents a strong candidate for PET imaging of diseases linked to ghrelin receptor overexpression.
Ipamorelin-Induced Insulin Secretion in Normal and Diabetic Rats
Administration of ipamorelin elicited a statistically significant increase (p<0.04) in pancreatic insulin secretion in both normoglycemic and diabetic rats. This stimulatory effect was markedly attenuated (p<0.03) following pretreatment with diltiazem, yohimbine, propranolol, or a combined regimen of atropine, propranolol, and yohimbine. Notably, atropine administration produced a significant reduction (p<0.007) in ipamorelin-induced insulin secretion in diabetic rats, whereas no such effect was observed in normoglycemic counterparts.
Protective Role of Ipamorelin in Glucocorticoid-Compromised Bone Formation
The study evaluated whether the growth hormone secretagogue ipamorelin could mitigate glucocorticoid-induced muscle and bone deterioration in adult rats. Eight-month-old female rats received daily methylprednisolone (9 mg/kg), ipamorelin (100 µg/kg, three times daily), or a combination of both for three months. Muscle strength was assessed via in vivo measurement of calf muscle tetanic tension, and bone formation was evaluated by periosteal activity. Co-administration of ipamorelin with glucocorticoids significantly improved maximum tetanic muscle force and enhanced periosteal bone formation—approximately four times greater than glucocorticoid treatment alone. These findings indicate that ipamorelin effectively counteracts glucocorticoid-induced declines in skeletal muscle strength and bone formation.
Referenced Citations
Andersen NB, Malmlöf K, Johansen PB, Andreassen TT, Ørtoft G, Oxlund H. The growth hormone secretagogue ipamorelin counteracts glucocorticoid-induced decrease in bone formation of adult rats. Growth Horm IGF Res. 2001 Oct;11(5):266-72. doi: 10.1054/ghir.2001.0239. PMID: 11735244.
Adeghate E, Ponery AS. Mechanism of ipamorelin-evoked insulin release from the pancreas of normal and diabetic rats. Neuro Endocrinol Lett. 2004 Dec;25(6):403-6. PMID: 15665799.
Peptidomimetic growth hormone secretagogue derivatives for positron emission tomography imaging of the ghrelin receptor



